LEARNING OBJECTIVES
- Recognize that the model of matter that we use during this unit is essentially that proposed by Democritus.
- Relate observations of diffusion to particle motion and collision in both liquid and gas phases.
- Relate observations regarding the addition of energy by warming to increased particle motion.
- Explain, at the particle level, how a thermometer measures the temperature of the system.
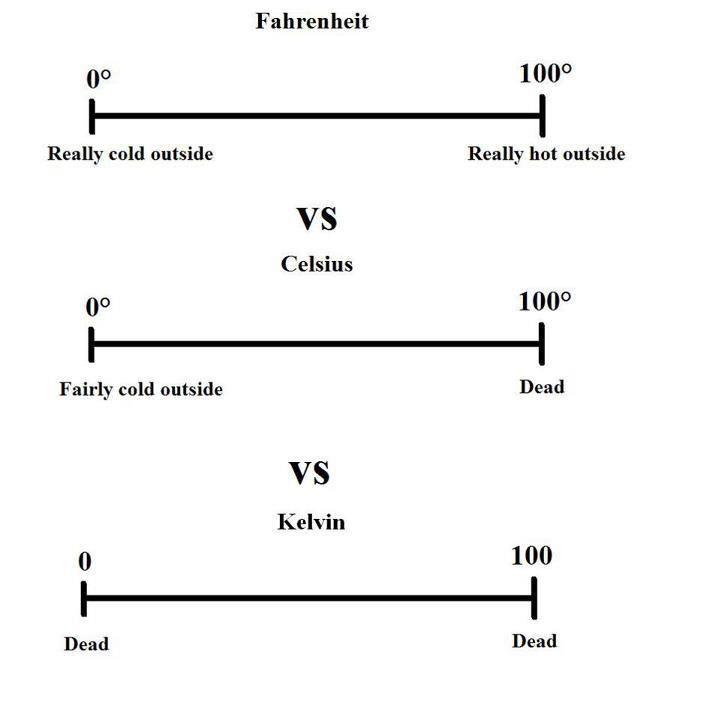
- Explain the basis for the Celsius temperature scale.
- State the basic tenets of Kinetic Molecular Theory (KMT) as they relate to gases. Particles of a gas:
- are in constant motion, moving in straight lines until they collide with another particle or a wall of the container in which they are enclosed.
- experience elastic collisions; i.e., they do not eventually “run down”
- do not stick to other particles.
- The speed of the particles is related to their temperature.
- The pressure of a gas is related to the frequency and impact of the collisions of the gas particles with the sides of the container in which they are enclosed.
- The 3 variables P, V and T are interrelated. Any factor that affects the number of collisions has an effect on the pressure. You should be able to:
- Predict the effect of changing P, V or T on any of the other variables.
- Explain (in terms of the collisions of particles) why the change has the effect you predicted.
- Explain the basis for the Kelvin scale. Keep in mind that one must use the absolute temperature scale to solve gas problems.
- Use factors to calculate the new P, V or T. Make a decision as to how the change affects the variable you are looking for.
NOTES
VIDEO
Structure of the Atom 1: The Earliest Models
Eureka Videos:
- Particles in Solid
- Particles in a Liquid
- Evaporation & Condensation
- Expansion & Contraction
- Measuring Temperature
- Temperature vs. Heat
The 1st Celsius Thermometer
The World’s Longest Straw
Pressure Conversions
Crush the Can Demo
Boyle’s Law
Charles’ Law
Gay-Lussac’s Law
Combined Gas Laws Practice Problems (2.4)
Ideal Gas Laws
Dalton’s Law (partial Pressure)
Phase Change Diagrams
Vapor Pressure
SIMULATIONS
Gizmo: Phase Changes
Boyle’s Law and Charles’s Law
pHet: Exploring Gas Properties
C-K12 Phase Diagrams
NetLogo Diffusion Simulator
WEBSITES
Pressure Concept Builder
Diffusion Activity